A paper in the Proceedings of the National Academy of Sciences (PNAS), “Kinematic self-replication in reconfigurable organisms” reports a novel form of self-replication, never seen in natural organisms, in synthetic organisms designed by artificial intelligence software and assembled from embryonic stem cells of the African clawed frog Xenopus laevis.
The paper begins by stating the significance of the work.
Almost all organisms replicate by growing and then shedding offspring. Some molecules also replicate, but by moving rather than growing: They find and combine building blocks into self-copies. Here we show that clusters of cells, if freed from a developing organism, can similarly find and combine loose cells into clusters that look and move like they do, and that this ability does not have to be specifically evolved or introduced by genetic manipulation. Finally, we show that artificial intelligence can design clusters that replicate better, and perform useful work as they do so. This suggests that future technologies may, with little outside guidance, become more useful as they spread, and that life harbors surprising behaviors just below the surface, waiting to be uncovered.
Here is the abstract.
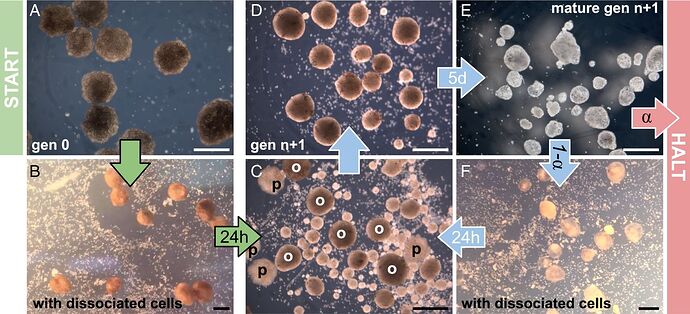
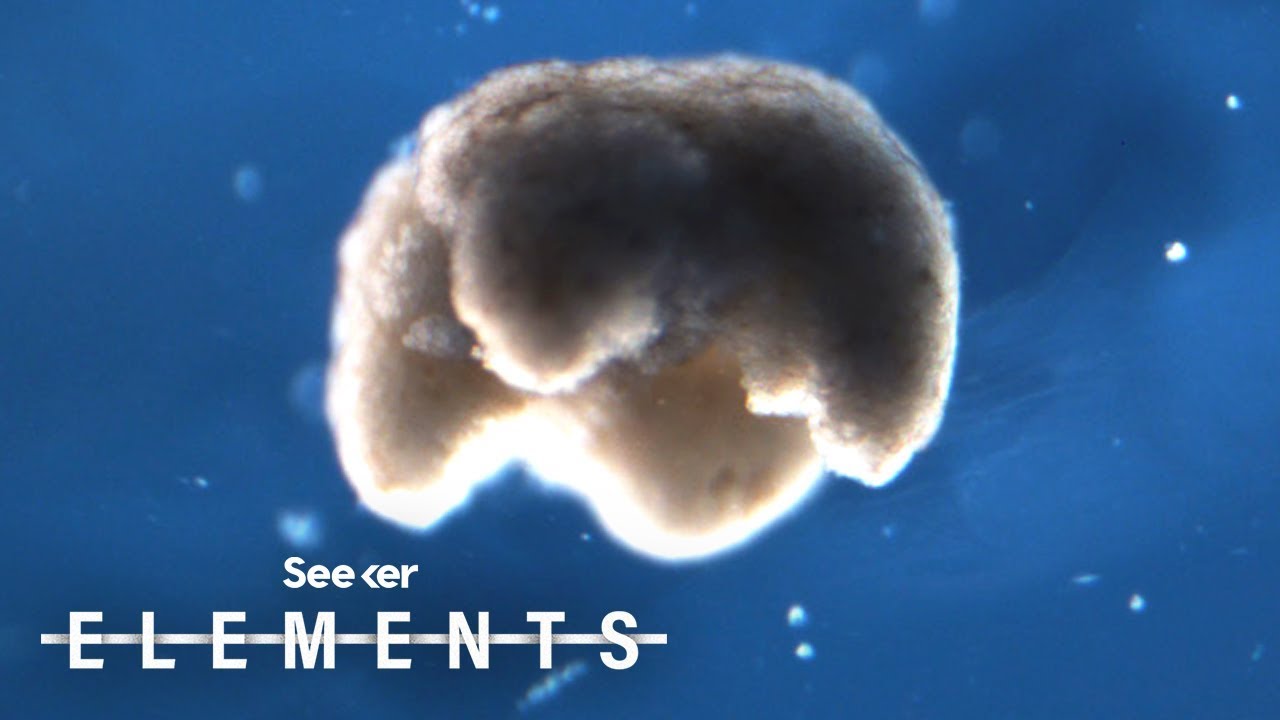
All living systems perpetuate themselves via growth in or on the body, followed by splitting, budding, or birth. We find that synthetic multicellular assemblies can also replicate kinematically by moving and compressing dissociated cells in their environment into functional self-copies. This form of perpetuation, previously unseen in any organism, arises spontaneously over days rather than evolving over millennia. We also show how artificial intelligence methods can design assemblies that postpone loss of replicative ability and perform useful work as a side effect of replication. This suggests other unique and useful phenotypes can be rapidly reached from wild-type organisms without selection or genetic engineering, thereby broadening our understanding of the conditions under which replication arises, phenotypic plasticity, and how useful replicative machines may be realized.
The Wyss Institute at Harvard University issued a popular description of the work, “Team builds first living robots—that can reproduce”.
“People have thought for quite a long time that we’ve worked out all the ways that life can reproduce or replicate. But this is something that’s never been observed before,” says co-author Douglas Blackiston, Ph.D., the senior scientist at Tufts University and the Wyss Institute who assembled the Xenobot “parents” and developed the biological portion of the new study.
“This is profound,” says [co-author Michael] Levin. “These cells have the genome of a frog, but, freed from becoming tadpoles, they use their collective intelligence, a plasticity, to do something astounding.” In earlier experiments, the scientists were amazed that Xenobots could be designed to achieve simple tasks. Now they are stunned that these biological objects—a computer-designed collection of cells—will spontaneously replicate. “We have the full, unaltered frog genome,” says Levin, “but it gave no hint that these cells can work together on this new task,” of gathering and then compressing separated cells into working self-copies.
“These are frog cells replicating in a way that is very different from how frogs do it. No animal or plant known to science replicates in this way,” says Sam Kriegman, Ph.D., the lead author on the new study.
⋮
“We asked the supercomputer at UVM to figure out how to adjust the shape of the initial parents, and the AI came up with some strange designs after months of chugging away, including one that resembled Pac-Man,” says Kriegman. “It’s very non-intuitive. It looks very simple, but it’s not something a human engineer would come up with. Why one tiny mouth? Why not five? We sent the results to Doug and he built these Pac-Man-shaped parent Xenobots. Then those parents built children, who built grandchildren, who built great-grandchildren, who built great-great-grandchildren.” In other words, the right design greatly extended the number of generations.“We’ve discovered that there is this previously unknown space within organisms, or living systems, and it’s a vast space,” says Bongard. “How do we then go about exploring that space? We found Xenobots that walk. We found Xenobots that swim. And now, in this study, we’ve found Xenobots that kinematically replicate. What else is out there?”
What could possibly go wrong? At least they didn’t name them “xenomorphs”.
If you’d like to try this, yourself, at home, the code is posted at GitHub. Bring your own frog.